How to reverse your type 2 diabetes
This guide gives you an overview of what you need to know about treating and reversing type 2 diabetes.1 Our other guides can teach you more about the symptoms of diabetes, as well as provide specific information about type 2 diabetes and type 1 diabetes.
Many people with diabetes or prediabetes have improved their health with dietary changes. You can too! Making these changes may allow you to reduce or eliminate diabetes medication, as well as help you lose weight.2
1. What is diabetes?
Simply put, diabetes is a disorder of blood sugar (glucose) and insulin. In diabetes, something is wrong with the way a person makes and/or uses insulin, a pancreatic hormone that lowers blood sugar by moving it out of the bloodstream and into the body’s cells.
Type 1 diabetes results when, for autoimmune or other rare reasons, the pancreas becomes damaged and fails to produce insulin. This form of diabetes is most often diagnosed in childhood but can occur in adults.
In type 2 diabetes, there are defects in both the production of insulin by the pancreas (insulin deficiency) and the use of insulin by the body (insulin resistance). When damage to the pancreas’ insulin-producing cells progresses to the point where the pancreas can no longer spontaneously release enough insulin to overcome the body’s resistance to it, blood sugar levels rise.
Excess glucose in the blood is a problem because it can damage blood vessels. What’s more, the body’s tissues can’t effectively use glucose for energy because too much of it stays in the bloodstream instead of entering the cells.
It is important to recognize that high glucose levels are a consequence of an underlying process that has been going on for years before blood sugar becomes high.
The good news is that diet and exercise can help decrease insulin resistance and its associated weight gain, which may help prevent or even put diabetes into remission.
To learn more about diabetes, click here:
2. Testing blood sugar
Our guide on what you need to know about blood sugar can help you learn more about both high and low blood sugar. This guide focuses specifically on the high blood sugar levels that occur in diabetes.
How do you know if you have too much sugar in your blood? If you don’t know already, it’s simple to test in a few seconds, either in your doctor’s office or with your own inexpensive blood glucose meter.6
Compare your own blood sugar reading with the ranges below:7
- Normal blood sugar: Less than 100 mg/dL (5.6 mmol/L ) after fasting overnight, and up to 140 mg/dL (7.8 mmol/L ) two hours after a meal
- Prediabetes: Between 100-125 mg/dL (5.6-7.0 mmol/L) after fasting overnight, or higher than 140 2 hours after a glucose tolerance test8
- Diabetes: 126 mg/dL (7.0 mmol/L) or higher after fasting overnight, or higher than 200 mg/dL (11.1 mmol/L) at any time
Keep in mind that you should not use glucometer readings alone to make a diagnosis of diabetes or prediabetes. If your blood sugar is high on a glucometer, ask your doctor to run a blood test to confirm the diagnosis. Also, most guidelines state that a single abnormal blood sugar reading is not sufficient to secure a diagnosis of diabetes; at least two are needed.
If you are already on a low carbohydrate diet and you are concerned about the measurements you’re getting, find out how a low carb diet affects blood sugar measurements.
To learn more about how to test your blood sugar, click here:
3. Food & diabetes
People with diabetes have difficulty keeping blood sugar levels in a normal range. The blood turns “too sweet” as glucose levels rise.9
Sugar in your blood comes from two places: your liver and the food that you eat. You can’t directly control the amount of sugar your liver makes, but you can control the foods you eat.
Foods are made up of three broad categories known as macronutrients (major nutrients): carbohydrate, protein, and fat. Many foods are a combination of two or all three macronutrients, but we often group foods according to whether they are mostly carbohydrate, protein, or fat.
Carbohydrates and blood sugar
Carbohydrates, or carbs, usually come from starches or sugars and turn into glucose when they are digested. When glucose enters the bloodstream, it’s called blood glucose, or blood sugar.

Carbohydrates
The more carbohydrate eaten in a meal, the more sugar is absorbed into the bloodstream and usually the higher the blood sugar will be.
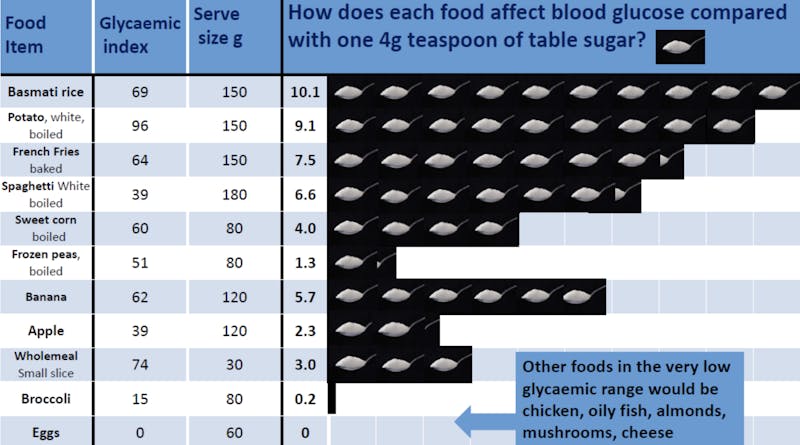
Although very few people would agree that sugary foods are good for you, some foods that we think of as “healthy” — such as fruit — can have a lot of sugar. And many people don’t know that starchy foods — such as bread, rice, pasta, and potatoes — quickly turn to sugar when you digest them.10
For some people, eating a potato could raise blood sugar as much as eating 9 teaspoons of sugar! It can be hard to predict exactly how someone’s blood sugar will respond, as this will likely vary based on genetics and baseline insulin sensitivity.11 By testing your blood sugar before eating and every 30-minutes after eating for up to two hours, you can quickly learn how different foods affect your blood glucose level. The results may surprise you!



Protein
Protein-containing foods include eggs, poultry, meat, seafood and tofu. Although individuals have different responses to these foods, consuming moderate amounts of protein at a meal generally has a mild to no effect on blood sugar.12
Fat
Dietary fat by itself has practically no effect on blood sugar. However, we seldom eat fat all by itself. Some foods, like cheese, are made up of mostly protein and fat. These foods probably won’t raise your blood sugar very much.13
But other foods, like doughnuts and French fries, are made up mostly of carbohydrate and fat. Because they’re high in carbs and fat together, these foods are likely to significantly raise your blood sugar.
4. How to lower blood sugar with diet
What happens if you remove foods that raise your blood sugar from your diet? Is there anything good left to eat? We think so. In fact, we have a whole guide on the best foods to control diabetes.
But a picture is worth a thousand words. These are just a few of the delicious foods that don’t raise blood sugar for just about everyone:




Many people with type 2 diabetes are now choosing a diet based primarily on low carb foods, and many clinicians are catching on as well.14
A person with type 2 diabetes will often notice that, starting with the first low carb meal, their blood sugar improves. The need for medications, especially insulin, is usually dramatically reduced. Substantial weight loss and health marker improvements often follow.15 Finally, people usually feel better and have more energy and alertness.16
Choosing foods low in carbs is an effective way to help you control your blood sugar and is safe for most people. However, if you are taking medications for your diabetes, you must work with your healthcare provider to adjust your medications when you change your diet since the need for medications, especially insulin, may be greatly reduced.17
Once you’ve consulted with your healthcare provider, you can try our 7-day meal plan for diabetes. We’ve done the groundwork for you and put together a collection of keto recipes that provide high amounts of protein and less than 20 grams of net carbs per day. You’ll be able to enjoy some of our most popular keto recipes, such as Egg bites with turkey bacon and Smash burgers with green beans.
If you are looking for a doctor who will work with you to control your diabetes with a change in diet, our map may help you find one.
5. The science of diabetes reversal
In 2019, the American Diabetes Association (ADA) stated that reducing carbohydrate intake was the most effective nutritional strategy for improving blood sugar control in those with diabetes.18
Research shows that low carb diets are a safe and effective option for treating type 2 diabetes. This body of evidence includes systematic reviews and meta-analyses of randomized controlled trials (the highest quality of evidence by our ratings.).19
A meta-analysis from 2017 found that low carb diets reduced the need for diabetes medication and also improved certain biomarkers in people with type 2 diabetes. This included reductions in hemoglobin A1c (HbA1c), triglycerides, and blood pressure; and increases in high-density lipoprotein (HDL) cholesterol, sometimes called the “good” cholesterol.20
Additionally, in a non-randomized trial from Virta Health, the intervention group of subjects with type 2 diabetes followed a very low carb diet and received remote monitoring by physicians and health coaches. After one year, 94% of those in the low carb group had reduced or stopped their insulin use. Furthermore, 25% had an HgbA1c in the normal range without needing any medications, suggesting their disease was in remission, and an additional 35% did the same with only metformin.21
At the two-year mark, a high proportion of subjects continued to demonstrate sustained improvements in glycemic control.22
Other interventions have also demonstrated efficacy for inducing remission of type 2 diabetes, although there is a lack of consistency with how different trials define “remission.”23 The DiRECT trial reported severe caloric restriction (eating around 850 calories per day) resulted in 46% remission at one-year.24 And bariatric surgery demonstrates between 25% and 50% diabetes remission up to ten-years post surgery.25
This evidence suggests that type 2 diabetes does not have to be a progressive and irreversible disease. It is clearly a treatable disease.
6. A message of hope
As recently as 50 years ago, type 2 diabetes was extremely rare. Now, around the world, the number of people with diabetes is increasing rapidly and is heading towards 643 million by 2030. This is a worldwide epidemic.
In the past, type 2 diabetes was thought to be a progressive disease with no hope for reversal or remission. People were — and sometimes still are — taught to “manage” type 2 diabetes, rather than to try to reverse the underlying process.
But now people with type 2 diabetes can hope to regain their health! Today we know that the hallmarks of type 2 diabetes — high blood sugar and high insulin — can often be reversed with a very low carb diet, severe caloric restriction, or weight loss surgery.
People don’t just have to “manage” their diabetes as it progresses. Instead, they can often lower their blood sugar to normal levels with diet alone, and may be able to avoid or discontinue most medications.
Normal blood sugar levels and fewer or no medications likely means no progression of disease, and no progression of complications. People with a diagnosis of type 2 diabetes may be able to live long, healthy lives, with toes, eyesight, and kidneys intact!
If you are not on any medications, you can start your journey back to health today. If you are on medications for diabetes or for other conditions, consult your doctor before beginning any lifestyle change, such as a low carb diet, so your medications are adjusted safely as your blood sugars improve.
When you’re ready, here’s where to start: A low carbohydrate diet for beginners. During your own journey, you might be inspired by some spectacular diabetes success stories.
How to reverse your type 2 diabetes - the evidence
This guide is written by Adele Hite, RD and was last updated on June 19, 2025. It was medically reviewed by Dr. Michael Tamber, MD on February 7, 2022.
The guide contains scientific references. You can find these in the notes throughout the text, and click the links to read the peer-reviewed scientific papers. When appropriate we include a grading of the strength of the evidence, with a link to our policy on this. Our evidence-based guides are updated at least once per year to reflect and reference the latest science on the topic.
All our evidence-based health guides are written or reviewed by medical doctors who are experts on the topic. To stay unbiased we show no ads, sell no physical products, and take no money from the industry. We're fully funded by the people, via an optional membership. Most information at Diet Doctor is free forever.
Read more about our policies and work with evidence-based guides, nutritional controversies, our editorial team, and our medical review board.
Should you find any inaccuracy in this guide, please email andreas@dietdoctor.com.
Some disagree with the use of the word “reverse” when it comes to type 2 diabetes. The concern is that it implies the disease is completely gone, never to return. At Diet Doctor, we use the term “reverse” to indicate that blood sugar levels are no longer in the diabetic range without the use of medications. However, we acknowledge that high glucose levels will likely recur if a patient goes back to their prior high-carb eating habits. Therefore, “reverse” does not imply a forever cure. ↩
The European Journal of Clinical Nutrition 2017: The interpretation and effect of a low carbohydrate diet in the management of type 2 diabetes: a systematic review and meta-analysis of randomised controlled trials [strong evidence]
Frontiers in Endocrinology 2019: Long-term effects of a novel continuous remote care intervention including nutritional ketosis for the management of type 2 diabetes: a 2-year non-randomized clinical trial [non-randomized trial; weak evidence] ↩
Centers for Disease Control and Prevention: Type 2 Diabetes ↩
This is based on statistics from the CDC. ↩
Journal of Clinical Endocrinology and Metabolism 2001: Clinical review 135: The importance of beta-cell failure in the development and progression of type 2 diabetes. [overview article; ungraded]
Frontiers in Endocrinology 2013: Beta cell dysfunction and insulin resistance. [overview article; ungraded]
↩Diet Doctor will not benefit from your purchases. We do not show ads, use any affiliate links, sell products or take money from industry. Instead we’re funded by the people, via our optional membership. Learn more ↩
These definitions are based on the ADA guidelines ↩
A glucose tolerance test involves drinking a standardized glucose solution and then testing blood sugar 2 hours later. ↩
In fact, prior to blood glucose tests being available, doctors would sometimes (hopefully not too often) diagnose diabetes by tasting sweet urine. Thankfully, we now have reliable, easily accessible blood meters instead. ↩
Studies have consistently shown that all digestible carbs have a much greater impact on blood sugar levels than protein or fat:
Diabetes Care 2004: Dietary carbohydrate (amount and type) in the prevention and management of diabetes [overview article; ungraded] ↩
PLOS Biology 2018: Glucotypes reveal new patterns of glucose dysregulation [observational study, weak evidence] ↩
Diabetes 2004: Effect of a high-protein, low-carbohydrate diet on blood glucose control in people with type 2 diabetes [randomized trial; moderate evidence]
American Journal of Clinical Nutrition 2003: An increase in dietary protein improves the blood glucose response in persons with type 2 diabetes [randomized trial; moderate evidence] ↩
The trials included in this review of RCTs did not restrict protein or fat intake and showed significant improvement in blood glucose levels and metabolic health:
BMJ Open Diabetes Research and Care: Systematic review and meta-analysis of dietary carbohydrate restriction in patients with type 2 diabetes [strong evidence]
↩Here are some amazing stories from some of those people: Diabetes success stories
And here is a list of medical doctors recommending a low carb or ketogenic diet
↩Diabetes Research and Clinical Practice 2018: Effect of dietary carbohydrate restriction on glycemic control in adults with diabetes: A systematic review and meta-analysis [strong evidence]
BMJ Open Diabetes Research and Care 2017: Systematic review and meta-analysis of dietary carbohydrate restriction in patients with type 2 diabetes [strong evidence]
Diabetes, Obesity & Metabolism 2019: An evidence‐based approach to developing low‐carbohydrate diets for type 2 diabetes management: a systematic review of interventions and methods [strong evidence] ↩
This is based mainly on clinical experience of low carb practitioners and was unanimously agreed upon by our low carb expert panel [weak evidence]. You can learn more about our panel here.
However, there is one small randomized controlled trial that reported these findings as well.
Diabetes Rsearch and Clinical Practice 2014: Randomization to a low carbohydrate diet advice improves health related quality of life compared with a low-fat diet at similar weight-loss in Type 2 diabetes mellitus[randomized trial; moderate evidence] ↩
Diabetes Therapy 2018: Effectiveness and safety of a novel care model for the management of type 2 diabetes at 1 year: an open-label, non-randomized, controlled study [weak evidence] ↩
Diabetes Care 2019: Nutrition therapy for adults with diabetes or prediabetes: a consensus report [overview article; ungraded] ↩
Nutrition Reviews 2022: Effect of a very low carbohydrate ketogenic diet vs recommended diets in patients with type 2 diabetes: a meta-analysis [strong evidence]
International Journal of Endocrinology 2021: Comparing the efficacy and safety of low carbohydrate diets with low-fat diets for type 2 diabetes mellitus patients: a systematic review and meta-analysis of randomized clinical trials [strong evidence]
Diabetes Research and Clinical Practice 2018: Effect of dietary carbohydrate restriction on glycemic control in adults with diabetes: A systematic review and meta-analysis [strong evidence]
BMJ Open Diabetes Research and Care 2017: Systematic review and meta-analysis of dietary carbohydrate restriction in patients with type 2 diabetes [strong evidence]
Diabetes, Obesity & Metabolism 2019: An evidence‐based approach to developing low‐carbohydrate diets for type 2 diabetes management: a systematic review of interventions and methods [strong evidence]
However, even meta-analyses of RCTs can have issues with scientific quality as we explained in our post on the following publication.
BMJ 2021: Efficacy and safety of low and very low carbohydrate diets for type 2 diabetes remission: systematic review and meta-analysis of published and unpublished randomized trial data [systematic review of randomized trials; strong evidence]
↩European Journal of Clinical Nutrition 2017: The interpretation and effect of a low carbohydrate diet in the management of type 2 diabetes: a systematic review and meta-analysis of randomised controlled trials [strong evidence] ↩
Diabetes Therapy 2018: Effectiveness and safety of a novel care model for the management of type 2 diabetes at 1 year: an open-label, non-randomized, controlled study [weak evidence] ↩
Frontiers in Endocrinology 2019: Long-term effects of a novel continuous remote care intervention including nutritional ketosis for the management of type 2 diabetes: A 2-year non-randomized clinical trial [weak evidence] ↩
According to the American Diabetes Association, remission means maintaining normal blood sugar in the absence of glucose-lowering medications for at least 3 months. As described in the following review, the definition of remission has not been consistent across all studies.
PLoS Medicine 2020: Defining remission of type 2 diabetes in research studies: A systematic scoping review[overview article; ungraded] ↩
In this trial, the investigators defined remission as more than 2 months of an HbA1c of less than 6.5% off glucose lowering medications. By this definition, people with blood sugar still in the pre-diabetes range (5.7-6.4%) were included in their remission statistics.
Lancet 2018: Primary care-led weight management for remission of type 2 diabetes (DiRECT): an open-label, cluster-randomised trial[moderate evidence]
A 2-year analysis of the same study calculated an expected cost-saving for payors.
Diabetologia 2020: Type 2 diabetes remission: 2 year within-trial and lifetime-horizon cost-effectiveness of the Diabetes Remission Clinical Trial (DiRECT)/Counterweight-Plus weight management programme [mathematical estimation; very weak evidence]
↩In th following RCT, they defined remission as 1-year of an HbA1c less than 6.5% off all glucose-lowering medications.
Lancet 2021: Metabolic surgery versus conventional medical therapy in patients with type 2 diabetes: 10-year follow-up of an open-label, single-centre, randomised controlled trial[moderate evidence]
Journal of Endocrinology and Metabolism 2020: Diabetes Remission Status During Seven-year Follow-up of the Longitudinal Assessment of Bariatric Surgery Study [nonrandomized study, weak evidence] ↩



